 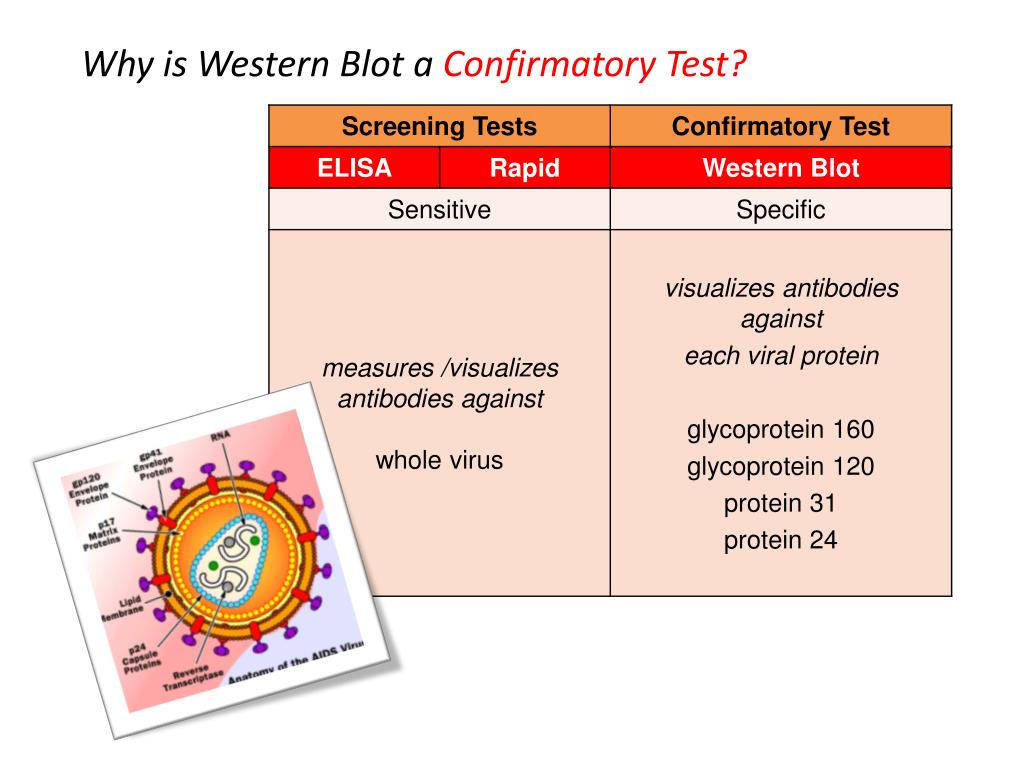
In conclusion, INNO and Geenius had comparable test performance, although the ease of use and shorter assay time for Geenius may make it the preferred choice for laboratories. Hence, further testing by a nucleic acid test or a p24 antigen test of specimens reactive on screening with a fourth generation Ag/Ab assay that are negative on confirmatory testing for HIV-specific antibody, may be useful. 
Nevertheless, overall poor sensitivity (25.5% - 44.7%) of these assays for the detection of early cases was observed, likely because these cases had very low or non-detectable levels of HIV antibodies. When early HIV cases were considered, INNO and Geenius were more likely to confirm an early-stage infection as positive. However, less non-specific reactivity was observed with the INNO and Geenius assays, where both of them were able to resolve MP-WB indeterminate cases. We show that all 3 assays had comparable test sensitivity in the detection of HIV-1 positive cases. Using the HIV Blot 2.2 (MP-WB), the INNO-LIA HIV I/II Score (INNO), and the Geenius HIV 1/2 Confirmatory Assay (Geenius), we tested 199 HIV-1 positive, 161 HIV negative, 65 HIV western blot indeterminate, 26 HIV seroconversion, 34 early HIV infection and 4 HIV-2 positive archived specimens. In view of recent revised recommendations for human immunodeficiency virus (HIV) confirmatory testing, the performance of 3 HIV confirmatory assays was compared. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
